Liberty professors team up to develop, refine world’s smallest ventilator
August 7, 2020 : By Ted Allen - Office of Communications & Public Engagement

Albert Einstein once said, “The definition of genius is taking the complex and making it simple.”
That is the marching order that Dr. Brian Walsh, a health sciences professor and director of Liberty University’s respiratory therapy program, gave to a team of volunteer engineers, physicians, and public health advocates from across the country and as far as South Africa in virtual conferences this spring.
Their mission was to design a compact, disposable ventilator that can be mass-produced on a global scale to assist medical workers treating patients in crises such as the COVID-19 pandemic.
The team applied principles of fluidics — a technique that uses fluids or gases to perform functions similar to electronic devices — to create an oscillator triggered by air pressure. (Fluidics were studied intensely during the Space Race in the 1960s but quickly died out when microprocessors were invented in the early 1970s).
“By basically reinventing and repurposing old technology, our understanding of gas laws and fluidics, combined with new deployable manufacturing techniques, we have developed a viable ventilator,” Walsh said of the small, plastic device that fits in the palm of his hand. “It can work off of the flow of air, takes no electricity, has no moving parts, and can be mass produced quickly by using 3D printing or computer numerical control machining operations.”
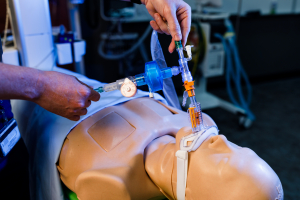
Walsh, who launched Liberty’s respiratory therapy program in the department of Allied Health Professions last summer, tested a prototype of the device with assistance from School of Engineering Professor Dr. Wayne Strasser.
“We brought in Dr. Strasser to create computer simulations for in-care patient environments to make sure this device continues to be safe, even in field hospitals where conditions are uncontrolled,” Walsh said. “We can’t mimic all of those gas conditions on the lung simulator we have (in the respiratory therapy lab) on campus, so that’s why we go to the computers to speed up our discovery and identify potential device vulnerabilities.”
“I study what particles and flows are doing on a microscopic level,” Strasser added. “Dr. Walsh is more knowledgeable of the safety of the patient, and he does amazing work. My job is to figure out how and why it works. This is the world’s smallest device to help unconscious patients to ventilate, and we’ve designed it to keep the patient happy and safe.”
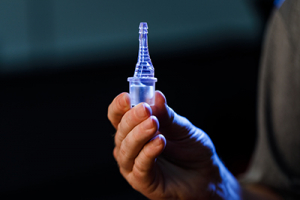
Walsh said the device uses the Coandă effect, which causes the ventilator to cycle between the patient’s inspiration (inhalation) and expiration (exhalation) without moving parts. (Lack of parts is what kept manufacturers from being able to mass-produce the ventilators needed at the beginning of the COVID-19 pandemic.) A simple three-step color-coded system makes it easy for the medical providers to determine precise pressure levels needed based on the patient’s breathing rate, which indicates when to escalate or de-escalate ventilatory support.
“We made the monophonic (single transmission path) devices to cover a wider patient population with no adjustments,” Walsh said. “This allows those with less experience or education to operate easily in a disaster situation where experts are hard to come by.”
Drs. Strasser and Walsh have already made major contributions to the medical field related to COVID-19 this year. Starting in March, Strasser used his expertise in computational fluid dynamics (CFD) and Walsh used his understanding of breathing patterns to construct computer models of COVID-19-infected patients for Vapotherm, Inc., assisting the company in testing its patented high-velocity respiratory therapy equipment to better contain the spread of the virus in hospitals.

Though he has 36 U.S. patents for inventions in engineering over the past 25 years, Strasser had no previous experience in the medical field, but has expanded his horizons to meet the urgent need.
“I’m available for whatever the Lord has for me to do,” he said. “It is an amazing group, no doubt about it.”
Strasser and Walsh have joined the team in establishing a public benefit corporation, Hospital Overload Protocol for Emergencies (HOPE), in order to obtain Food and Drug Administration (FDA) approval for their design, the HOPE inVent ventilator. The device utilizes trademarked FluidIQ valve technology featured in a line of products that are easily scalable and cost-effective, filling gaps in emergency preparedness protocols. Walsh is optimistic the ventilator will receive premarket approval from the FDA by the end of September.
The team is also seeking a grant from the National Institutes of Health to develop a mobile phone app to supplement the device.
Walsh said the project has the potential to revolutionize respiratory therapy, and he feels privileged to have joined such a synergistic team in developing a life-saving device.
“It’s been a wonderful experience and helps us at Liberty to live out our mission and vision, particularly during this crisis,” Walsh said. “It has allowed us to establish a relationship with this group of engineers who will continue to develop this product, and there are plenty of future uses for students and faculty throughout this process as we advance this technology.”


